Key Market Insights
Leveraging their expertise, over 450 types of kits, media, reagents and extracellular matrices have been manufactured by consumable providers for research and therapeutic purposes
The market features the presence of more than 80 firms across the globe; majority of these stakeholders are emerging players based in North America
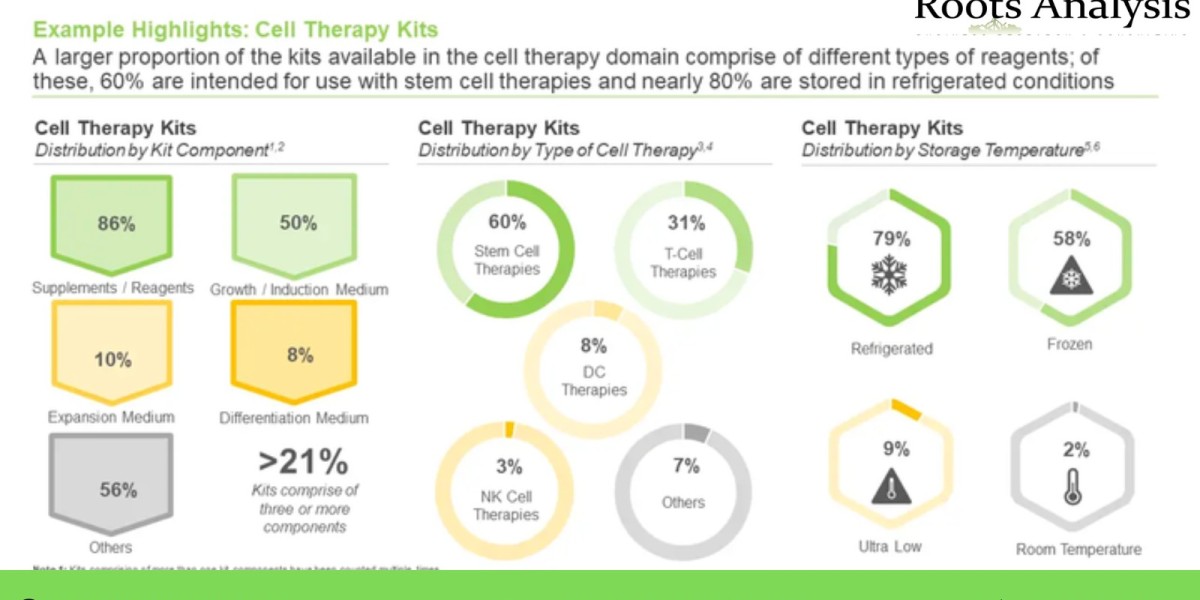
A larger proportion of the kits available in the cell therapy domain comprise of different types of reagents; of these, 60% are intended for use with stem cell therapies and nearly 80% are stored in refrigerated conditions
Media developers are offering products for a broad range of cell therapies; nearly 90% of such players are providing media in the volume range of 100 to 500 ml
Predominantly, the cell therapy reagents are intended to be used at discovery scale of operation for a wide spectrum of functions, including cell expansion and proliferation
The market landscape of matrices, which typically have a shelf life of 1 to 1.5 years, is well distributed in terms of type of ECM coating and type of formulation
In pursuit of gaining a competitive edge, cell therapy consumable providers are upgrading their existing technologies and expanding their product portfolios
Many companies have undertaken strategic initiatives, including partnerships, acquisitions and expansions, to augment their existing capabilities
We expect industry stakeholders to continue to forge strategic alliances with niche / specialized players engaged in this domain to further augment their respective product offerings
Over the years, stakeholders within this industry have established strong brand positions by undertaking a range of initiatives to further advance the development of raw materials for cell therapies
Cost is a key determinant for the adoption of consumables in a cell therapy manufacturing process; the pricing strategy matrix is likely to assist players in evaluating the competitive market prices for their products
In 2035, the commercial scale of operation is likely to account for 75% of the total demand for cell therapy consumables; this is attributed to the expected surge in the anticipated approvals of multiple cell therapies
A paradigm shift from animal-based to animal component free formulations, combined with stringent regulatory guidelines, is likely to drive the growth of the cell therapy consumables market at an annualized rate of 12%
The projected future opportunity for cell therapy consumable providers is expected to be well distributed across different types of end-users and key geographical regions
Table of Contents
1. PREFACE
1.1. Introduction
1.2. Key Market Insights
1.3. Scope of the Report
1.4. Research Methodology
1.5. Frequently Asked Questions
1.6. Chapter Outlines
2. EXECUTIVE SUMMARY
3. INTRODUCTION
3.1. Context and Background
3.2. Introduction to Cell Therapies
3.3. Comparison of Cell Therapies with Other Biopharmaceuticals
3.4. Classification of Cell Therapy Products
3.5. Overview of Cell Therapy Development and Manufacturing
3.6. Role of Raw Materials in Cell Therapy Development and Manufacturing
3.7. Types of Cell Therapy Consumables
3.8. Key Challenges Associated with Manufacturing of Cell Therapy Consumables
3.9. Future Perspectives
4. MARKET LANDSCAPE
4.1. Chapter Overview
4.2. List of Cell Therapy Kit Providers
4.3. List of Cell Therapy Media Providers
4.4. List of Cell Therapy Reagent Providers
4.5. List of Cell Therapy Extracellular Matrix Providers
5. COMPANY COMPETITIVENESS ANALYSIS
5.1. Chapter Overview
5.2. Key Assumptions and Parameters
5.3. Methodology
5.4. Cell Therapy Consumables Providers: Company Competitiveness Analysis
5.5. Cell Therapy Kits Providers
5.6. Cell Therapy Media Providers
5.7. Cell Therapy Reagent Providers
5.8. Cell Therapy Extracellular Matrix Providers
6. BRAND POSITIONING OF KEY INDUSTRY PLAYERS
6.1. Chapter Overview
6.2. Scope and Methodology
6.3. Brand Positioning: STEMCELL Technologies
6.4. Brand Positioning: Miltenyi Biotec
6.5. Brand Positioning: Thermo Fisher Scientific
6.6. Brand Positioning: Takara Bio
6.7. Brand Positioning: GeminiBio
7. COMPANY PROFILES
7.1. Chapter Overview
7.2. STEMCELL Technologies
7.2.1. Company Overview
7.2.2. Product Portfolio
7.2.3. Recent Developments and Future Outlook
7.3. Miltenyi Biotec
7.4. Thermo Fisher Scientific
7.5. Bio-Techne
7.6. Irvine Scientific
7.7. Lonza
7.8. Sartorius
7.9. BD Biosciences
7.10. Corning
7.11. CellGenix
8. RECENT DEVELOPMENTS AND INITIATIVES
8.1. Chapter Overview
8.2. Partnership Models
8.3. Cell Therapy Consumables: Partnerships and Collaborations
8.4. Cell Therapy Consumables: Mergers and Acquisitions
8.5. Cell Therapy Consumables: Recent Expansions
9. LIKELY PARTNER ANALYSIS FOR CELL THERAPY CONSUMABLE PROVIDERS
9.1. Chapter Overview
9.2. Scoring Criteria and Key Assumptions
9.3. Scope and Methodology
9.4. Key Potential Strategic Partners for Cell Therapy Consumable Providers
10. ROOTS ANALYSIS PRICING STRATEGY
10.1. Chapter Overview
10.2. Roots Analysis Framework
11. DEMAND ANALYSIS
11.1. Chapter Overview
11.2. Scope and Methodology
11.3. Global Demand for Cell Therapy Consumables
11.4. Analysis by Scale of Operation
11.5. Analysis by Geography
12. MARKET FORECAST AND OPPORTUNITY ANALYSIS
12.1. Chapter Overview
12.2. Key Assumptions and Methodology
12.3. Global Cell Therapy Consumables Market, 2023-2035
12.4. Cell Therapy Consumables Market: Analysis by Type of Product
12.5. Cell Therapy Consumables Market: Analysis by Type of Cell Therapy
12.6. Cell Therapy Consumables Market: Analysis by Scale of Operation
12.7. Cell Therapy Consumables Market: Analysis by Type of End-User
12.8. Cell Therapy Consumables Market: Analysis by Geography
13. UPCOMING TRENDS AND FUTURE GROWTH OPPORTUNITIES
13.1. Chapter Overview
13.2. Emerging Trends Related to Cell Culture Media
13.3. Automation of Cell Therapy Manufacturing Processes
13.4. Single Use Systems and Technologies in Cell Therapy Manufacturing
14. CONCLUDING REMARKS
15. INTERVIEW TRANSCRIPTS
16. APPENDIX I: TABULATED DATA
17. APPENDIX II: LIST OF COMPANIES AND ORGANIZATIONS
To view more details on this report, click on the link
https://www.rootsanalysis.com/reports/cell-therapy-consumables-market.html
You may also be interested in the following titles:
Nanoparticle Formulation Market |
About Roots Analysis
Roots Analysisis a global leader in the pharma / biotech market research. Having worked with over 750 clients worldwide, including Fortune 500 companies, start-ups, academia, venture capitalists and strategic investors for more than a decade, we offer a highly analytical / data-driven perspective to a network of over 450,000 senior industry stakeholders looking for credible market insights.
Contact:
Ben Johnson
+1 (415) 800 3415